Unit 1: CHEMEIA OF THE BIOS
B-PRION READING: 08-18-14
A study on prions revealed that there were major discrepancies between prions in their natural and lab made states. Natural prions are extremely infectious, while test tube prions aren't until made to do so, but not quite as dangerous, they are the cause of nuerodegenerative disease, and studies have provided no solutions to these complexities.
B-Coacervates Reading: 08-25-14
This article was about rapid RNA exchange in two-phase system. The goal of the experiment that took place was to determine the ability of ATPs and coacervate droplets to retain RNA, similar to fatty acid-based vesicles. To achieve these results, it took many different tests, chemicals, and graphs. In the article it also spoke of how they planned to decrease the exchange rate between the RNA and the coacervate droplets specifically. The RNA would only stay on any particular droplet for a few seconds. They wanted to do this because as the droplets spread near and far, they would not be heritable. Understanding how all of these interact and work, combined with knowledge about fatty acids and phospholipid vesicles, it may lead to multiple possibilities for the development of earlier cells, and even how to cure the incurable diseases we have running amuck though society.
B-PRION READING: 08-18-14
A study on prions revealed that there were major discrepancies between prions in their natural and lab made states. Natural prions are extremely infectious, while test tube prions aren't until made to do so, but not quite as dangerous, they are the cause of nuerodegenerative disease, and studies have provided no solutions to these complexities.
B-Coacervates Reading: 08-25-14
This article was about rapid RNA exchange in two-phase system. The goal of the experiment that took place was to determine the ability of ATPs and coacervate droplets to retain RNA, similar to fatty acid-based vesicles. To achieve these results, it took many different tests, chemicals, and graphs. In the article it also spoke of how they planned to decrease the exchange rate between the RNA and the coacervate droplets specifically. The RNA would only stay on any particular droplet for a few seconds. They wanted to do this because as the droplets spread near and far, they would not be heritable. Understanding how all of these interact and work, combined with knowledge about fatty acids and phospholipid vesicles, it may lead to multiple possibilities for the development of earlier cells, and even how to cure the incurable diseases we have running amuck though society.
G-LAB 8: PINEAPPLE ENZYMES & JELLO MOLDS: 08-15-14
PROCEDURE:
1. gather all supplies:
-3 beakers
-10 test tubes (5 per trial)
-hot plate
-frozen, fresh, canned and cooked pineapple
- jello
-stirring rods and knives
2. Follow the instructions on the jello box to make jello.
3. pour jello (liquid) into the test tubes (one inch)
4. Put one piece of each kind of pineapple in each kind of test tube (leave last test tube with out pineapple)
5. Let sit until jello forms
6. retrieve jello and record results
7. repeat steps 2-6 for second trial.
PROCEDURE:
1. gather all supplies:
-3 beakers
-10 test tubes (5 per trial)
-hot plate
-frozen, fresh, canned and cooked pineapple
- jello
-stirring rods and knives
2. Follow the instructions on the jello box to make jello.
3. pour jello (liquid) into the test tubes (one inch)
4. Put one piece of each kind of pineapple in each kind of test tube (leave last test tube with out pineapple)
5. Let sit until jello forms
6. retrieve jello and record results
7. repeat steps 2-6 for second trial.
S-Pattern Matching: 08-28-14
G-Presence or Absence of Enzymes: 08-28-14
In this lab, no reactions occurred. We contribute this to many factors, as it could have been weak solutions, expired solutions, inconcise timing, or even human error. Multiple things might have gone wrong, but the fact remains that there were no reactions to occur.
In this lab, no reactions occurred. We contribute this to many factors, as it could have been weak solutions, expired solutions, inconcise timing, or even human error. Multiple things might have gone wrong, but the fact remains that there were no reactions to occur.
Toothpickase Activity
Time (seconds)
Number of Toothpicks Broken
10: 5
20:12
30: 20
60: 41
120: 50
180: 50
360: 50
1. A. In this model, what is the substrate? Unbroken toothpicks
B. What acts as the enzyme? The person
C. What is the Protein? Broken toothpicks
2. The fastest time period for the reaction rate was 180 seconds.
3. As the toothpick supply runs out the speed stays the same.
4. The reaction rate would slow as the substrate became less concentrated.
5. If more toothpicks were added, the reaction rate would decrease.
6. We could increase the enzyme concentration by adding more people, and as such the reaction rate would increase.
7. With a competitive inhibitor you would most likely go faster, increasing the reaction rate, and as for a noncompetitive inhibitor the reaction rate would stay the same. To represent this, we could add more people for the competitive inhibitor, and just go together for the noncompetitive inhibitor.
Macromolecules Lab
Carbon-Based Molecule
Positive Test Color
Negative Test Color
Protein
Darker Blue
Lighter Blue
Sugar
Darker Blue
Lighter Blue
Starch
Dark Green
Brown/Orange
Lipid
Transparent
Nontransparent
Unknowns
Protein
Lipid
Starch
Sugar
Starch
No RXN: Dark Blue
No RXN: Nontransparent
RXN: Black
No RXN: Blue
Sugar
No RXN: Dark Blue
No RXN: Nontransparent
No RXN: Yellow
RXN: Green
1. The Data we collected was qualitative, as we did not do multiple trials, thus taking extra caution.
2. Two limitations to the indicators that we used were:
-can add more iodine to make bluer
-only one color
3. We could improve any errors made, by making it a quantitative lab, as well as using more than one type of indicatior.
Conclusion
The unknowns have been deduced to be starch and sugar. We know that because of the reactions made, as well as the colors that the compounds turned. They went from yellowish to brownish black, and from blue to a light green. Starch and sugar are both carbohydrates, and as thus will give us quick short term bursts of energy. We also feel that we used polysaccharides for the carbohydrates to give us a more intricate reaction.
Enzyme Lab
Part A
What gas is being released? Oxygen
Rate of reaction. 1
Has it gotten colder or warmer? Colder
Is it endothermic or exothermic? Exothermic
What is the liquid composed of? Water and liver particles
What do you think would happen if you added more liver to this liquid? No reaction, nothing left to react with
Second reaction rate. 0
Is catalase reusable? Explain. No, because all of the reactants have already been used.
Part B
Substance
First rate of reaction
Second rate of reaction
Potato
0
0
Apple
0
0
Chicken
1
1
Which tissues contained catalase? Catalase
Do some contain more catalase than others? How can you tell? Yes, because the reactions are faster or slower.
Part C
What will boiling do to an enzyme? It will denature it.
What is the reaction rate for the boiled liver and peroxide? 0
What is the reaction rate for the cold liver/peroxide? 0
What is the reaction rate for the warm liver/peroxide? 0
Part D
Rate of Reaction for: Strong Acid 4 Acid 2 Strong Base 0 Base 0
What is the optimal pH for catalase (estimate)? 1-6
Part E
For a new experiment, I would take multiple types of food, both hard and soft, as well as canned versus uncanned. I, then, would use different chemicals, such as H2SO4 and KOH to see the new reactions. I would then place them all in test tubes, to see said reactions and record the results.
Data Analysis
1. Catalase breaks down hydrogen peroxide. The enzyme is the catalase, and the peroxide is the substrate. When the reaction takes place the catalase takes the hydrogen peroxide bonds and breaks them apart.
2. No, during Part A we took used liver and put it in more peroxide. No reaction took place.
3. Temperature and pH can cause the enzyme to denature, affecting the substrate’s reaction, and add more room for testing your hypothesis.
4. First: Get raw and processed sugars, as well as artificial sweeteners.
Second: Acquire a good amount of amylase and Benedict’s solution.
Third: Add the sugar to the test tubes, and proceed to add the amylase and solution.
Fourth: Set a timer and wait for a color change.
Fifth: Record the time it took, and what the colors were.
Time (seconds)
Number of Toothpicks Broken
10: 5
20:12
30: 20
60: 41
120: 50
180: 50
360: 50
1. A. In this model, what is the substrate? Unbroken toothpicks
B. What acts as the enzyme? The person
C. What is the Protein? Broken toothpicks
2. The fastest time period for the reaction rate was 180 seconds.
3. As the toothpick supply runs out the speed stays the same.
4. The reaction rate would slow as the substrate became less concentrated.
5. If more toothpicks were added, the reaction rate would decrease.
6. We could increase the enzyme concentration by adding more people, and as such the reaction rate would increase.
7. With a competitive inhibitor you would most likely go faster, increasing the reaction rate, and as for a noncompetitive inhibitor the reaction rate would stay the same. To represent this, we could add more people for the competitive inhibitor, and just go together for the noncompetitive inhibitor.
Macromolecules Lab
Carbon-Based Molecule
Positive Test Color
Negative Test Color
Protein
Darker Blue
Lighter Blue
Sugar
Darker Blue
Lighter Blue
Starch
Dark Green
Brown/Orange
Lipid
Transparent
Nontransparent
Unknowns
Protein
Lipid
Starch
Sugar
Starch
No RXN: Dark Blue
No RXN: Nontransparent
RXN: Black
No RXN: Blue
Sugar
No RXN: Dark Blue
No RXN: Nontransparent
No RXN: Yellow
RXN: Green
1. The Data we collected was qualitative, as we did not do multiple trials, thus taking extra caution.
2. Two limitations to the indicators that we used were:
-can add more iodine to make bluer
-only one color
3. We could improve any errors made, by making it a quantitative lab, as well as using more than one type of indicatior.
Conclusion
The unknowns have been deduced to be starch and sugar. We know that because of the reactions made, as well as the colors that the compounds turned. They went from yellowish to brownish black, and from blue to a light green. Starch and sugar are both carbohydrates, and as thus will give us quick short term bursts of energy. We also feel that we used polysaccharides for the carbohydrates to give us a more intricate reaction.
Enzyme Lab
Part A
What gas is being released? Oxygen
Rate of reaction. 1
Has it gotten colder or warmer? Colder
Is it endothermic or exothermic? Exothermic
What is the liquid composed of? Water and liver particles
What do you think would happen if you added more liver to this liquid? No reaction, nothing left to react with
Second reaction rate. 0
Is catalase reusable? Explain. No, because all of the reactants have already been used.
Part B
Substance
First rate of reaction
Second rate of reaction
Potato
0
0
Apple
0
0
Chicken
1
1
Which tissues contained catalase? Catalase
Do some contain more catalase than others? How can you tell? Yes, because the reactions are faster or slower.
Part C
What will boiling do to an enzyme? It will denature it.
What is the reaction rate for the boiled liver and peroxide? 0
What is the reaction rate for the cold liver/peroxide? 0
What is the reaction rate for the warm liver/peroxide? 0
Part D
Rate of Reaction for: Strong Acid 4 Acid 2 Strong Base 0 Base 0
What is the optimal pH for catalase (estimate)? 1-6
Part E
For a new experiment, I would take multiple types of food, both hard and soft, as well as canned versus uncanned. I, then, would use different chemicals, such as H2SO4 and KOH to see the new reactions. I would then place them all in test tubes, to see said reactions and record the results.
Data Analysis
1. Catalase breaks down hydrogen peroxide. The enzyme is the catalase, and the peroxide is the substrate. When the reaction takes place the catalase takes the hydrogen peroxide bonds and breaks them apart.
2. No, during Part A we took used liver and put it in more peroxide. No reaction took place.
3. Temperature and pH can cause the enzyme to denature, affecting the substrate’s reaction, and add more room for testing your hypothesis.
4. First: Get raw and processed sugars, as well as artificial sweeteners.
Second: Acquire a good amount of amylase and Benedict’s solution.
Third: Add the sugar to the test tubes, and proceed to add the amylase and solution.
Fourth: Set a timer and wait for a color change.
Fifth: Record the time it took, and what the colors were.

Unit 2: Cells
LAB 4: MEETING THE "PROTISTS": 09-12-14
1A. Plasmodium spp.
LAB 4: MEETING THE "PROTISTS": 09-12-14
1A. Plasmodium spp.
- The life cycle stage is the Trophozoite Stage.
1B. The Cilliates
Paramecium Caudatum and Vorticella
Identify: Macronucleus, Oral Groove, Cillia, Food Vacuole, Contractile Vaculoe
Macronucleus: Found in Vorticella around the center, the large circles that are dark green around the edges.
Cillia: Found in paramecium, The very light colored rim surrounding the cell.
Oral Groove: Found in Parimicium, extremely difficult to see in the photo, located between the 2 sun shapes (Contractile Vacuole)
Food Vacuole: Found in Paramecium, large yellow circle to the left of the Contractile Vacuole at the bottom of the cell.
Contractile Vacuole: Found in Paramecium, large Sun shaped organelles in the cell.
Paramecium Caudatum
Vorticella
1C. The Oomycetes (Water Molds or Egg Molds)
Saprolegnia spp.
Identify: Zoosporangia, Oogonium, Antheridium
Saprolegnia
Zoosporangia
Oogonium
Antheridium
Part 2: The Plantae
2A. The Chlorophytes (Green Algae)
Chlamydomonas spp., Spirogyra spp., Volvox spp., Ulva spp.
Identify:
Chlamydomonas: Stigma, Chloroplasts
Stigma: Very small black dot, located near the flagella of the cell
Chloroplasts: Small Light colored circles within the cells, located throughout.
Spirogyra: Spiral Chloroplasts
Spiral Chloroplasts: All the little green dots that make a spiraling pattern throughout the cell.
Volvox: Cells, Daughter Colony
Cells: the very tiny dots covering the entire volvox, very light in color.
Daughter Colony: All of the very dark green medium sized circles within the cell.
Ulva: Gametophytes, Sporophytes, Zoospores
Gametophytes: (definition) an individual or generation of plants exhibiting alterations of generations that bear sex organs.
Sporophytes: (definition) an individual or generation of plants exhibiting alterations of generations that bear asexual spores.
Zoospores: (definition) a motile flagellated asexually produced spore.
Chlamydomonas
Volvox
Spirogyra
Ulva
Part 3: The Excavates
3A. The Diplomonads
Giardia Lamblia
Giardia Lamblia
3B. The Kinetoplastids
Trypanosoma Gambiense
Identify: Nucleus, Flagella
Nucleus: Around the center of the dark purple worm like cells. The darkest spot of purple in them.
Flagella: The thinnest end of the worm like cells.
Trypanosoma
Part 4: Rhizaria
4A. The Foraminiferans
Foraminifera Strew
Foraminifera Strewn
4B. The Radiolarians
Radiolaria Strew
Radiolaria Strew
Part 5: Unikonts
5A. The Laboseans
Amoeba Proteus
Amoeba
5B. The Plasmodial Slime Molds
Dictydium spp.
Identify: Plasmodium, Sporangium
Dictydium
Plasmodium
Sporangia
5C. The Cellular Slime Molds
Identify: Amoebas, Pseudoplasmodium, Fruiting Body
Amoebas
Pseudoplasmodium
Fruiting Body (Sorocarp)
Part 6: Recognizing Protists "In the Wild"
6A.
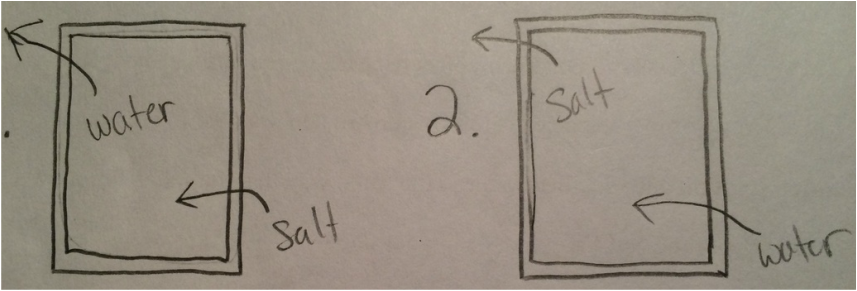
Osmosis/Plasmosis Lab Activity
3. The fact of being that is it is a plant cell, not an animal cell, where plants need to be submerged in water to stay alive.
4. It keeps them hydrated, so they don't shrivel up and die.
5.it kills the plants, as the salt steals all of the water out of the cells.
6.it will dehydrate yourself, thus making them unable to work.
7.The sugar is acting the same way as salt, thus absorbing the water trying to reach equilibrium.
Alcoholic Fermentation in Yeast
Glucose + Oxygen --- Carbon Dioxide + Water + Oxygen
1. Yeast is a microscopic unicellular fungus.
2. It is sugar, a carbohydrate.
3. Anaerobic conditions
Alcoholic Fermentation produces: carbon dioxide and alcohol
4.
4. The independent variable is the amount of yeast/The sugar solution
5. The 0% sucrose concentration with no yeast/to compare results
8a) It creates more ATP than anaerobic
8b) It doesn't need air
9. No, the 1% solution created the most carbon dioxide in 20 minutes.
10. Over time, the higher the concentration, more carbon dioxide is produced.
11. The group we talked to had similar results to ours.
Experiment 2
1. Yeast breaks down starch molecules into simple sugars and exudes a liquid that releases carbon dioxide and ethyl alcohol into existing air bubbles into the dough.
2. 10% because it releases the most amount of carbon dioxide causing the bread to rise.
3.How does cinnamon affect the bread?
4. We believe there will be no reaction except for the flavor.
5. The Independent Variable is the Water and Yeast, the dependent is the cinnamon.
6. The amount of sucrose
7. A) Fill test tubes with equal amounts of water
B) Add 10% sucrose
C) Add 0%, 1%, 5%, 10% cinnamon
D) Place a balloon on top of each test tube as you add the cinnamon
E) Test 10 min, 20 min, and 40 min
F) Record results of baloon and bubbles of CO2
8.
10. No, they didn't produce different amounts, they did match our hypothesis.
11. Our results mean, it only affects the taste and wont affect its raising.
Packet
Unit 3:Plants
4. It keeps them hydrated, so they don't shrivel up and die.
5.it kills the plants, as the salt steals all of the water out of the cells.
6.it will dehydrate yourself, thus making them unable to work.
7.The sugar is acting the same way as salt, thus absorbing the water trying to reach equilibrium.
Alcoholic Fermentation in Yeast
Glucose + Oxygen --- Carbon Dioxide + Water + Oxygen
1. Yeast is a microscopic unicellular fungus.
2. It is sugar, a carbohydrate.
3. Anaerobic conditions
Alcoholic Fermentation produces: carbon dioxide and alcohol
4.
4. The independent variable is the amount of yeast/The sugar solution
5. The 0% sucrose concentration with no yeast/to compare results
8a) It creates more ATP than anaerobic
8b) It doesn't need air
9. No, the 1% solution created the most carbon dioxide in 20 minutes.
10. Over time, the higher the concentration, more carbon dioxide is produced.
11. The group we talked to had similar results to ours.
Experiment 2
1. Yeast breaks down starch molecules into simple sugars and exudes a liquid that releases carbon dioxide and ethyl alcohol into existing air bubbles into the dough.
2. 10% because it releases the most amount of carbon dioxide causing the bread to rise.
3.How does cinnamon affect the bread?
4. We believe there will be no reaction except for the flavor.
5. The Independent Variable is the Water and Yeast, the dependent is the cinnamon.
6. The amount of sucrose
7. A) Fill test tubes with equal amounts of water
B) Add 10% sucrose
C) Add 0%, 1%, 5%, 10% cinnamon
D) Place a balloon on top of each test tube as you add the cinnamon
E) Test 10 min, 20 min, and 40 min
F) Record results of baloon and bubbles of CO2
8.
10. No, they didn't produce different amounts, they did match our hypothesis.
11. Our results mean, it only affects the taste and wont affect its raising.
Packet
Unit 3:Plants
Floating Leaf Disk Photosynthesis Lab: 10-23-14
Purpose: The purpose is to see how different light intensity effect photosynthesis.
Predictions:
How will the different light conditions affect the rate of floating disks?
The disks in the light will have the most floating disks, the disks in ambient light will have some, and disks in the dark will have none.
Purpose: The purpose is to see how different light intensity effect photosynthesis.
Predictions:
How will the different light conditions affect the rate of floating disks?
The disks in the light will have the most floating disks, the disks in ambient light will have some, and disks in the dark will have none.
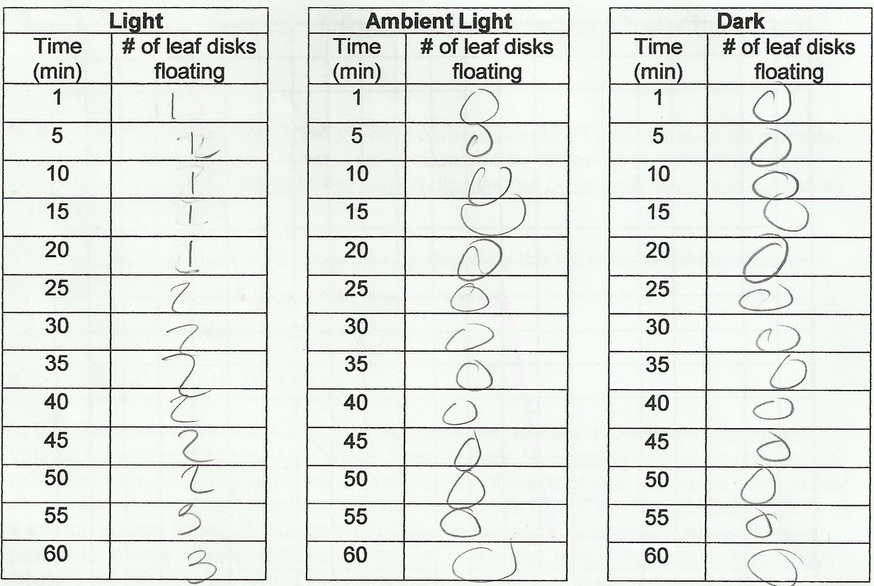
Qualitative Observations: The disks in the light had some disks floating and had soap bubbles. The disks in both ambient light and in the dark had none floating and no soap bubbles.
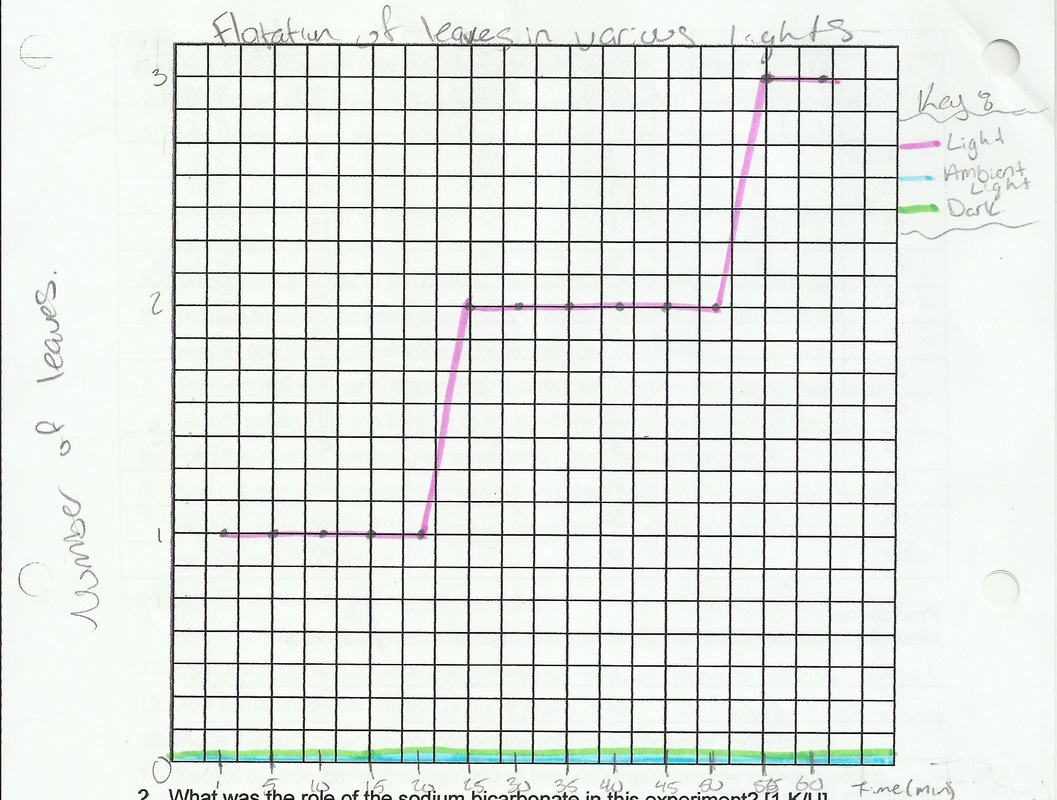
1. Graph your results for all three trials in the graph paper below. Label the graph, both axes and provide a legend to distinguish each trial.
2. What was the role of the sodium bicarbonate in this experiment?
1. Graph your results for all three trials in the graph paper below. Label the graph, both axes and provide a legend to distinguish each trial.
2. What was the role of the sodium bicarbonate in this experiment?
3. Which trial resulted in all the leaf disks floating the fastest? Explain.
4. Explain the process that caused the leaf disks to rise.
5. If the leaf disks were boiled, what kind of results would you expect?
6. How does light intensity affect the rate of photosynthesis?
7. The same experiment was conducted where 10 leaf disks were placed in a sodium bicarbonate solution and placed in the light. Every minute, the number of floating disks were counted and recorded. After 14 minutes, the leaf disks were floating.
4. Explain the process that caused the leaf disks to rise.
5. If the leaf disks were boiled, what kind of results would you expect?
6. How does light intensity affect the rate of photosynthesis?
7. The same experiment was conducted where 10 leaf disks were placed in a sodium bicarbonate solution and placed in the light. Every minute, the number of floating disks were counted and recorded. After 14 minutes, the leaf disks were floating.